By Kristen Mitchell
George Washington University experts at the forefront of the COVID-19 pandemic response have one resounding message: The best vaccine you can get is the first one available to you. In the shadow of 515,000 deaths in the United States, they said, this is no time to shop around.
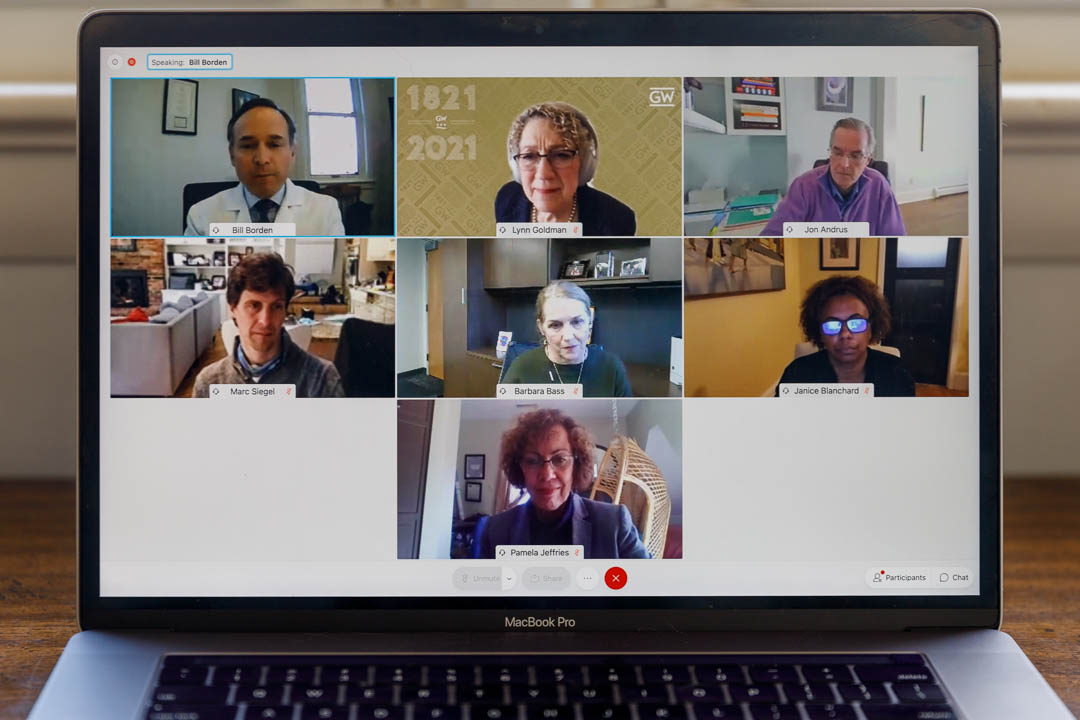
Researchers, clinicians and deans from the Milken Institute School of Public Health, the School of Medicine and Health Sciences and the School of Nursing spoke to the GW community on Tuesday as part of a discussion on COVID-19 vaccines—what the differences are between them, why they are safe, and why social distancing and masking remains critical post-shot.
The virtual event featured Lynn Goldman, the Michael and Lori Milken Dean of Public Health; SMHS Dean Barbara L. Bass, vice president for health affairs and CEO of the GW Medical Faculty Associates (MFA); GW Nursing Dean Pamela Jeffries; Jon Andrus, adjunct professor of global health; Janice Blanchard, professor of emergency medicine; Marc Siegel, associate professor of medicine; and Bill Borden, professor of medicine and chief quality and population health officer at the MFA.
Dr. Goldman, who moderated the event, called the imperative to vaccinate in the United States and around the world “one of the most important public health endeavors of our generation.”
Dr. Bass and Dr. Jeffries applauded the work of GW’s frontline workers while acknowledging the strain and trauma nurses, doctors and other healthcare workers have faced around the world. They have served the community while facing great personal peril, particularly early on in the pandemic when little was known about how to treat COVID-19 and supplies and resources were limited.
“The activities and energy and passion of this GW community in battling this disease, whether it’s in our clinics or in our hospital environments or in our schools, has been absolutely astonishing,” Dr. Bass said.
The U.S. Food and Drug Administration has authorized three vaccines for emergency use. The Pfizer-BioNTech and Moderna vaccines, which require two injections, and the newly approved Johnson & Johnson vaccine, which requires only one. The Pfizer-BioNTech and Moderna vaccines, which need to be stored at extremely low temperatures, both use messenger RNA (mRNA), which teaches the body’s cells how to make a protein that triggers an immune response. While this technology is new to the public, scientists have been studying it for years.
“I’m very, very confident that this is a safe technology,” said Dr. Siegel, senior co-investigator of GW’s Moderna Phase 3 vaccine trial. “It is new, but it has been in the research field for a while. And that’s why we were able to make the vaccine so quickly— we already had the manufacturing platforms that we needed to be able to produce this vaccine at scale.
If it had never been even thought of before, it would have taken a lot longer to start producing the vaccines that we have today.”
The Johnson & Johnson vaccine uses DNA instead of mRNA to provoke an immune response against COVID-19, similar to the annual flu shot. The vaccine is also more heat stable compared to the two other options, making it less challenging to store, said Dr. Andrus, an expert in epidemiology, vaccines, immunizations and response to emerging infectious disease threats.
“Logistically it’s a game changer,” he said. “It’s also just one dose, which those of you who’ve worked in the field know how much easier that makes it.”
More than 78 million doses of the COVID-19 vaccine have been administered so far in the United States, and President Joe Biden’s administration announced this week that every adult in the country should be vaccinated by the end of May. As vaccines become more widely available, it’s imperative that doses reach communities disproportionately harmed by this disease. Dr. Blanchard, a frontline health care worker, said community leaders need to ensure African Americans and other at-risk groups are getting vaccinated.
Dr. Blanchard posted about her positive experience getting the vaccine on Facebook. Old friends and acquaintances reached out to her afterward and told her the post made them confident about getting the vaccine when they are eligible. She recently wrote an opinion piece for CNN about how individuals can reverse community distrust by sharing their vaccine experiences with hesitant friends and family.
“I think the power of small communities and the power of telling other people is really important,” she said. “We can’t emphasize that [enough].”
Dr. Borden, who has been leading the MFA’s COVID-19 response and vaccine rollout, said some health care workers have been hesitant about receiving the vaccine. Mirroring national trends, hesitant frontline workers are often more open to getting the vaccine when they see trusted colleagues receive it.
Dr. Jeffries talked about the challenges elderly Americans face in registering for vaccine appointments. Individuals are required to register for appointments online in many regions, however, many elderly people do not use a computer. Her 85-year-old mother, who lives in a rural area, is scheduled to receive her second dose this week, but only after Dr. Jeffries was able to navigate the online registration process on her behalf. Many of her friends have been monitoring what their peers are doing.
“It can be an elaborate process for some if you’re not used to going through all the steps that need to be done,” she said.
As the country figures out the best way to deliver vaccines safely and quickly to as many people as possible, Dr. Borden said mass vaccination sites in areas that have been hit hard by COVID-19 are essential to curbing disparity in distribution. He applauded the work of frontline workers who provided free flu shots at Nationals Park on Election Day.
“One of the lessons that comes out of this is, we have to bring vaccinations, we need to bring health care, to where people live and where they go and lower those barriers to accessing care,” he said.
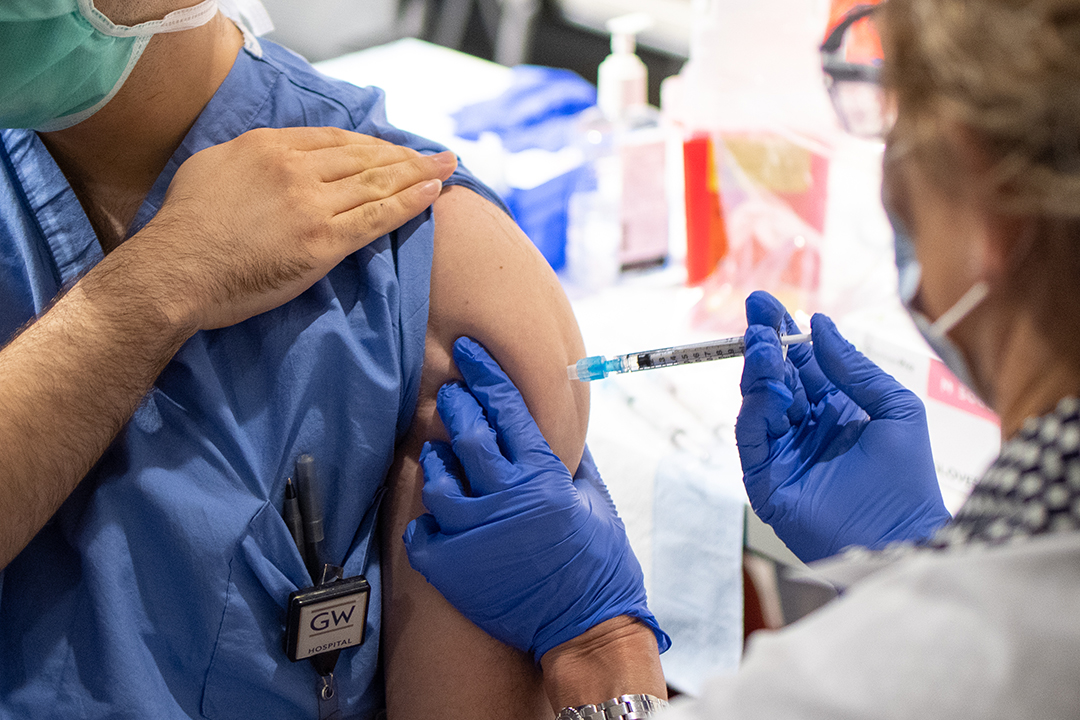
The panelists also spoke about their own experiences getting the vaccine, which included a range of minor symptoms such as soreness at the injection site, fatigue or no symptoms at all. Even after receiving the vaccine, individuals will need to continue day-to-day social distancing, mask wearing and good hygiene practices for the foreseeable future. It is not yet clear whether the vaccine prevents an individual from spreading COVID-19, even if they do not have symptoms.
Dr. Siegel said he was surprised by how emotional he felt receiving the shot.
“You really feel like there is some hope and some potential for a light at the end of the tunnel,” he said.
Dr. Andrus had a similar experience. He has heart disease, putting him at higher risk of serious COVID-19 complications. When he and his wife received their shots, they broke down in tears.
“It was very emotional, that here it’s unprecedented, the turnaround of this technology and science, and then to receive the benefit of that,” he said. “We had to jump a lot of hoops to wait for our turn, but it was very emotional.”
A recording of the COVID-19 Vaccines 101 event is available on GW’s Coronavirus website.