An arterial inflammatory disease called atherosclerosis is the United States’ leading cause of death, but about half of American adults over 45 have the condition and don’t know it, according to the National Institutes of Health. Characterized by plaque buildup inside the arteries, the vascular superhighways that push blood from the heart to the rest of the body, the condition often goes overlooked until it has become serious.
One promising solution may lie in a field that seems, to a layperson’s eye, unrelated: mechanical engineering. Nora Caroline Wild, a George Washington University graduate student in the School of Engineering and Applied Science, is using cardiovascular fluid dynamics to explore possible anatomical factors that could alert at-risk patients early.
“Once a patient has symptoms, that means they already have atherosclerosis which is typically pretty severe,” said Wild, a mechanical and aerospace engineering doctoral candidate in Department Chair Michael Plesniak’s Biofluid Dynamics Lab. “So I’m looking into what kind of pre-existing features could cause someone’s blood flow to have an unhealthy impact on the vessel tissue. Could we look at the geometry of a patient's blood vessels—the angle of their branching, the diameter—to statistically classify a patient as higher or lower risk?”
Plaque is composed of cholesterol, cellular waste and other fatty substances, all of which can accumulate on vascular walls as they are carried through the blood. But healthy endothelial cells, which line the walls of blood vessels, prevent the accumulation of plaque by a biochemical response to the flow of blood with which they’re in constant contact. Under ideal health conditions, the force of blood flow creates miniscule whirlpool-shaped vortices which exert a frictional force called shear stress against the arterial wall. Shear stress stimulates the endothelial cells, preventing plaque buildup before it can become dangerous.
But for some patients, Wild found, shear stress is affected by arterial geometry. Think of a hose spraying water up a ramp: If the ramp ends in a gentle slope back down to the ground, water will flow evenly up to the peak and down to the grass at its base. But if the ramp ends in a sharp 90-degree drop, pressurized water will arc over the grass at its base, barely wetting it. If hose, ramp and grass were all underwater, vortices would form as the pressurized flow pushed forward, applying shear stress to the grass under the gentle slope but little or none on the grass under the sharper drop.
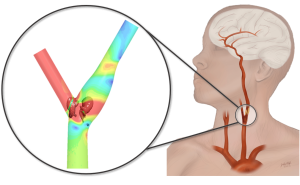
The common carotid artery, which is the critical vessel delivering blood to the face and brain, branches in an analogous way into the internal and external carotids at a spot in the neck known to be vulnerable to plaque buildup. Using patient-averaged clinical data, Wild created two models of this carotid bifurcation area. The first model, based on data from a healthy population, had a symmetric branching angle of 60 degrees—a gently sloping ramp over which blood flows evenly, creating effective, lasting vortices. The second model, based on anatomical features from patients with increased atherosclerosis risk, bifurcated asymmetrically and at a larger angle.
Running simulations of blood flow through this second model, Wild found that the critical vortices deteriorated at a faster rate. Not only were they pushed past the vulnerable junction by the sharp angle of bifurcation, but they didn’t last long enough to effectively whirl back. Untouched by the necessary shear stress, the walls of these vessels were more likely to retain buildup of plaque.
At these high-risk carotid junctions, blood flow functions “kind of like a D.C. summer,” Wild joked: you want a “nice little breeze” to keep yourself from drowning in sweat, but instead you’re oppressed by motionless air.
If Wild’s virtual findings translate clinically, patients could be noninvasively screened for atherosclerosis risk in a clinical setting by measuring the pressure gradients in their carotid. (Getting pressure gradients in arteries is possible in a clinical setting with sensors and novel imaging-based methods, Wild said.) Patients whose vascular geometry indicates increased danger could modify their behavior accordingly, including the establishment of a more active care plan.
“In the future, I believe this research will help to treat atherosclerosis before patients suffer severe symptoms,” Wild said. “We’re excited about these findings because they increase the understanding on why certain people are more prone for plaque formation and thus help identify clinical parameters for improved clinical early detection.”