Researchers at George Washington University have developed an experimental device that could one-day help monitor and treat heart disease and dysfunction in the days, weeks or months following traumatic heart-related event before dissolving safely in the body. The dissolvable device would also help avoid complications and burdens associated with removal from the body, such as infection or the high cost of another surgical procedure.
Nearly 700,000 people in the United States die from heart disease every year, according to the Centers for Disease Control and Prevention, and one in three people die from complications in the first weeks or months following a traumatic heart-related event.
GW researchers working with counterparts at Northwestern University describe the device they developed and tested in a new study published last week in Science Advances. Luyao Lu, assistant professor of biomedical engineering at GW’s School of Engineering and Applied Science and corresponding author of the study, said many of the deaths that occur following heart surgery or a heart attack could be preventable if doctors had better tools to monitor and treat patients in the delicate weeks after these events take place.
“The tool developed in our work has great potential to address the unmet needs in many programs of fundamental and translational cardiac research,” Lu said.
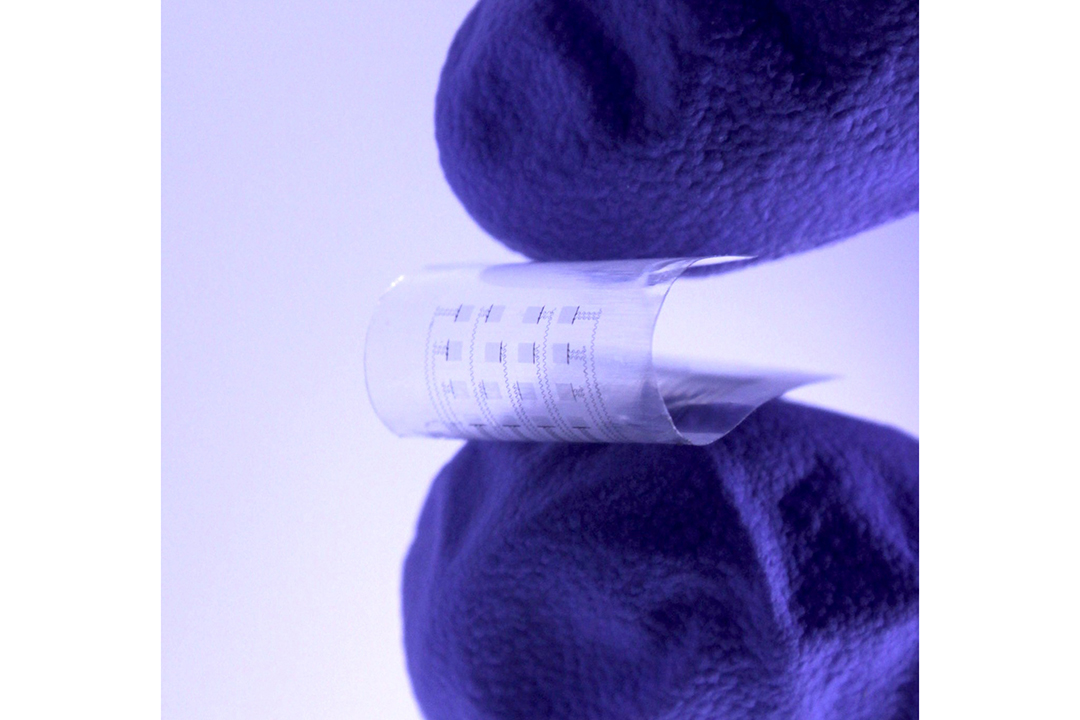
The device is an advanced soft electronics system that uses an array of sensors and actuators to perform more complicated investigations of the heart than traditional devices like a pacemaker has been able to do in the past. Not only can it be placed on various sections of the heart, but it can communicate information to physicians in real time about the health status of the patient’s heart.
In addition, the device is made of materials approved by the U.S. Food and Drug Administration that are compatible with the human body and able to fully dissolve after a clinically relevant period. The device is also highly transparent, allowing physicians to observe specific heart regions to make a diagnosis or provide treatment.
According to the researchers, the tool could potentially be used in any situation where short-term monitoring and control of parameters like cardiac electrical wave propagation or heart rate are needed. If additional testing pans out, the device could one day be used in temporary settings to restore normal heart rhythm.
Current devices such as temporary pacemakers can monitor the heart for a short period but eventually require surgical removal. This device completely dissolves into benign products in the body. The study’s authors say this feature could avoid complications from surgical extraction, lower infection risks and eliminate additional financial burdens for patients.
Zhiyuan Chen, a Ph.D. student at GW and lead study author, said materials that can dissolve in the body are more challenging to process because of their sensitivity to environmental factors like heat, moisture and chemicals.
“However, we developed a new manufacturing technology capable of achieving nanoscale dissolvable patterns,” Chen said. “This technology has the potential to advance the miniaturization and large-scale integration of dissolvable electronic systems, enabling new possibilities for broader and more diverse patient care and therapeutic functionalities.”
The study’s authors say this experimental device is not intended to replace a pacemaker, as it acts differently. While a pacemaker is meant to monitor and stimulate the heart long-term, this device is designed for temporary use when monitoring and treatment may only be required for a short period.
A pacemaker can also only provide one overall picture of the heart: whether the heart is beating. This device, the researchers say, provides a more nuanced picture that can show which parts of the heart are functioning well and which areas are not. In addition, the transparency of the device allows researchers to optically map many important cardiac physical parameters to better study heart function and heart disease mechanisms.
According to Igor Efimov, an experimental cardiologist and professor of biomedical engineering at Northwestern, several serious complications can follow cardiac surgeries or catheter-based therapies, including atrial fibrillation and heart block.
Current post-surgical monitoring and treatment of these complications require more sophisticated technology than is currently available. For example, temporary wires are inserted after surgery and connected to an external current generator to provide electric stimulation during temporary heart block caused by the surgery.
“Recently, we developed a bioresorbable pacemaker that will replace such a wire. Post-operative atrial fibrillation requires a more complicated approach based on a multi-electrode array for sensing and terminating atrial fibrillation,” Efimov said. “In this new study, we present novel technology to achieve such a goal: to provide a transient electronic device that will map electrical activity from numerous locations on the atria and will deliver electrical stimuli from many locations to terminate atrial fibrillation as soon as it starts.”
Although the researchers tested the device in the lab in rodents, future studies will have to prove it is safe and effective before it can be approved for use in a clinical setting. The researchers also say they’ll be exploring more ways in which this device could be used and altered for other medical needs where temporary monitoring is required.
The study was supported by the National Science Foundation and the National Institutes of Health.