Researchers at the George Washington University, whose members include the physician who diagnosed D.C.’s first AIDS patient in 1981, are once again at the forefront of the fight against the disease, thanks to a $7.5 million grant from the National Institutes of Health.
The five-year grant will fund the District’s first Center for AIDS Research (CFAR), a prestigious designation awarded to fewer than 20 others in the United States.
The goals of the D.C. CFAR include advancing HIV research in the District, funding new HIV investigators and increasing interdisciplinary collaborations among scientists and community partners.
Two of the center’s primary research areas will be preventing the spread of HIV among highly impacted populations and focusing on finding a cure for the virus.
The CFAR brings together nearly 200 researchers from three GW schools and five D.C. institutions: the Columbian College of Arts and Sciences, the Milken Institute School of Public Health, the School of Medicine and Health Sciences, American, Georgetown and Howard universities, as well as the Children’s National Medical Center and the Veterans Affairs Medical Center.
GW Professors Alan Greenberg, chair of the Department of Epidemiology and Biostatistics, and Gary Simon, the Walter G. Ross Professor of Medicine and of Microbiology and Tropical Medicine, will lead the center. It was Dr. Simon who diagnosed the first D.C. patient with AIDS in August 1981 at the George Washington University Hospital.
“We want to make D.C. a destination city for researchers who want to have a tangible impact on fighting the HIV epidemic,” Dr. Greenberg said. “The CFAR will have a strategic, proactive role in having stimulated that, and if there is ever a cure for HIV, why shouldn’t progress toward that goal come from a D.C.-based institution?”
The NIH created the CFAR program in 1988 with the goal to enhance and coordinate high-quality AIDS research projects. Rather than a brick and mortar center, a CFAR is a network of scientists and community partners all working in support of that mission. A CFAR provides the infrastructure and core services to recruit established researchers and build interdisciplinary collaborations, as well as funding and mentorship for new HIV investigators.
Before becoming a CFAR, a center receives preliminary funding to demonstrate its capabilities and build its infrastructure. The NIH awarded GW and its partner institutions $3.75 million in 2010 to create a developmental CFAR (D-CFAR). Now, five years later, Dr. Greenberg’s excitement for the full CFAR distinction is palpable.
“We thought that if the HIV research institutions in D.C. went in as a city-wide consortium,” Dr. Greenberg said, “then we could qualify as a CFAR and address the severity of the epidemic in the city,”
Funding the Next Generations of Researchers
A year after he diagnosed the first AIDS case in D.C., Dr. Simon and Allan Goldstein, both SMHS professors, began to investigate whether injecting high-risk patients with hormones from the thymus gland could prevent the onset of AIDS.
By 1984, scientists had identified HIV as the cause of the immune deficiency disease, but the first approved treatment for the virus did not become available until the end of the decade.
 |
| Gary Simon co-directs the D.C. Center for AIDS Research. |
“Throughout the ’80s it was terrible because virtually everyone died,” Dr. Simon said. “We were seeing more and more patients infected with the virus, and there was little that we could do for them that was effective.”
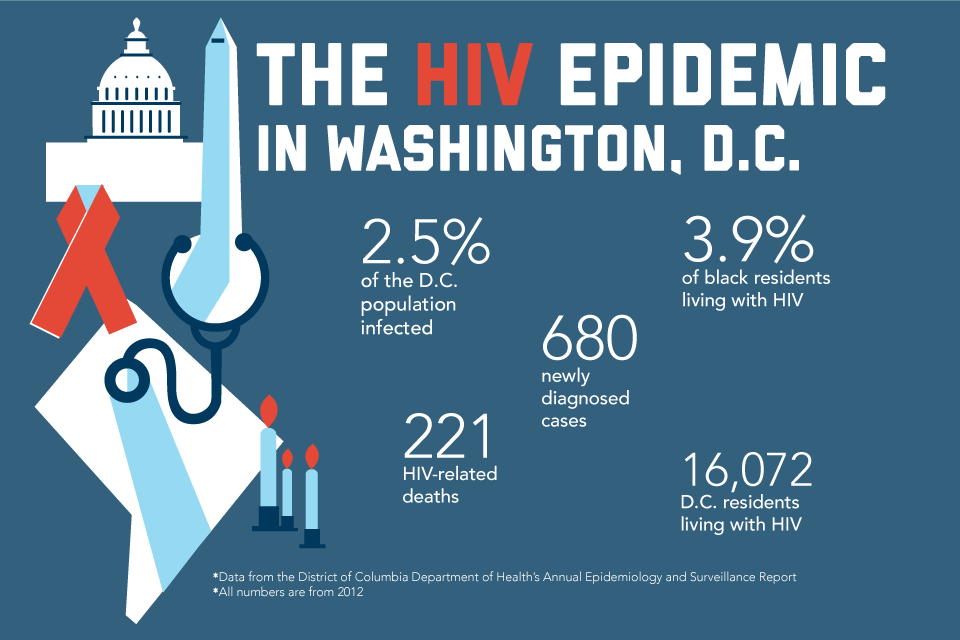
More than 30 years later, an HIV diagnosis is no longer a death sentence. The number of new cases in Washington, D.C., declined 42 percent from 2008 to 2012, according to the most recent data from the D.C. Department of Health. Still, HIV remains an epidemic in D.C., where more than 16,000 residents, or 2.5 percent of the population, are living with the virus. And so the search for a cure and a vaccine continues.
A large part of the D.C. CFAR will be to lay the groundwork for the next generation of investigators to help end the epidemic.
Since the developmental CFAR began in 2010, the program has awarded 30 pilot awards totaling $1.25 million to researchers. During that same time, members of the D-CFAR received 57 new NIH awards and 42 new HIV investigators were recruited to D.C. institutions. The newly-awarded $7.5 million NIH grant will double the amount of funding available to promote the development of early stage, women and minority HIV investigators in D.C.
These pilot awards are essential during a time when NIH funding is becoming increasingly competitive, said Jennifer Skillicorn, the administrative director of the CFAR.
“Junior investigators often find it difficult to get their first grant. It is the senior researchers with the most well-developed programs who keep getting funded,” she said. “The CFAR is a way to counter-balance that trend. It’s a really good way to jumpstart science and careers.”
In addition to providing funding for new research and connecting investigators once fragmented throughout the city, the CFAR aids in translating science from the lab to the community. The CFAR includes an active Community Advisory Board, which has representatives from community-based and nonprofit organizations throughout the city, such as Metro TeenAIDS, the Women’s Collective and Unity Health Care.
The board meets with Dr. Greenberg and CFAR leaders every three months to provide guidance, develop strategies to share research results with the community and participate in pilot award application reviews.
The community aspect of the center is key to its success, according to Doug Nixon, the chair of the Department of Microbiology, Immunology and Tropical Medicine. A renowned HIV researcher, Dr. Nixon joined SMHS in 2013 and directs the “basic sciences core” within the D.C. CFAR.
“Sometimes we scientists forget that we have to communicate with people about what we’re doing to make them as excited about our research as we are,” Dr. Nixon said. “The CFAR gives the community a scientific voice in the capital. I think that is pretty amazing.”
Searching for a Cure
After earning his medical degree at GW in 1982, Dr. Greenberg began his residency in Greenwich Village, at the height of the AIDS epidemic.
“That was a life-changing experience for me,” Dr. Greenberg said. “To honor the memory of so many of the people for whom we cared for, a lot of people, including myself, decided to commit our careers to the fight against HIV.”
Before returning to his alma mater as a faculty member in 2005, Dr. Greenberg spent 20 years at the Centers for Disease Control and Prevention, where he directed the HIV Epidemiology Branch from 1999 to 2005. During that time, the Food and Drug Administration approved AZT, the first antiretroviral drug for treating HIV/AIDS. It was a revolutionary event that began to change AIDS from a lethal disease to a chronic condition.
“Clinically, it’s been a rather dramatic journey,” said Dr. Simon who was working at GW in 1987 when the treatment was approved. “I can’t impress upon people how dramatic the cocktail was. Now I have HIV patients who I’ve been following for 20 years.”
While HIV patients on treatment can live relatively normal lives, they must remain in care. Linkage and retention to care, as well as adherence to medications are still problems in D.C., especially in communities affected by other health and socio-economic challenges, according to Dr. Greenberg. And an HIV patient’s life expectancy is, on average, five to 10 years shorter than an uninfected person.
“The epidemic is in no means over,” Dr. Greenberg said.
But the end might be in sight.
“Cure research,” led by Dr. Nixon, will be one of two scientific working groups in the D.C. CFAR.
The major barriers standing in the way of developing an HIV vaccine are the virus’s rapid infection and frequent mutations, Dr. Nixon said. Scientists have focused on neutralizing antibodies directed against HIV-1 to stop the virus, but the antibodies cannot keep up with virus’s replication.
In a study recently published in the journal Immunology, Dr. Nixon and a team of researchers found that a fossil virus—a dormant remnant of a retrovirus that lives in our DNA—could be a solution. Since fossil viruses do not change, the researchers believe they may be able to target a vaccine to the regions where they are expressed in HIV-infected cells.
These fossil viruses could also provide clues for an HIV cure. The challenge in cure research, Dr. Nixon said, is exterminating HIV-1 hiding in latent reservoirs of the body.
“We know that if a patient stops taking the drugs, the virus comes back. But we don’t know where the virus is hiding,” Dr. Nixon said. “So you either need to find where the virus is hiding or devise strategies that will attack the virus if it emerges.”
Dr. Nixon has already recruited new faculty for this endeavor, and his team is at the beginning stages of “very exciting” findings. The researcher does not particularly like the term “cure research,” but he, Dr. Simon and Dr. Greenberg—all who have already seen incredible transformations in HIV-related research during their lifetimes—are hopeful that progress can be made toward developing a vaccine and a cure.





