By Kristen Mitchell
George Washington University researchers received $3 million from the National Institutes of Health to test the efficacy of a hookworm vaccine candidate developed and tested at the university. This is the next step in their goal to fight hookworm, which infects millions of people worldwide every year.
The study is led by Jeffrey Bethony, professor of microbiology, immunology and tropical medicine in the School of Medicine and Health Sciences, and David Diemert, associate professor of microbiology, immunology and tropical medicine.
The research team will soon start phase two of clinical trials in which they will test the efficacy of a vaccine candidate on adult volunteers recruited in the Washington, D.C. area. The test is a “challenge study” to see whether the vaccine protects them from hookworm infection. Thirty-six volunteers will receive doses of the vaccine candidate, which has been tested for safety and immunogenicity both at GWU and in hookworm endemic sites in Brazil and Gabon under a Investigational New Drug application with the U.S. Food and Drug Administration. Twelve volunteers will receive placebo injections.
“This will be the first proof of concept or proof of efficacy study that we have done,” said Dr. Diemert. “Everything before has just been looking at immune responses and safety.”
Volunteers will receive three doses of the vaccine at two-month intervals. About a month after receiving the third injection the volunteers will return to the GW clinic to be infected with hookworm larvae that have been produced in John Hawdon’s lab, another GW researcher on the new grant. To transfer infection, researchers will pipet a sample of microscopic hookworm larvae onto a gauze pad, which will then be placed on the volunteer’s forearm for an hour. From there, the hookworm larvae penetrate the skin. For the next three months the researchers will monitor the volunteers for hookworm infection.
The test is a significant step forward in developing a vaccine to protect against hookworm, which affects more than 500 million people worldwide and can lead to anemia and impact cognitive and physical development, according to the Centers for Disease Control and Prevention.
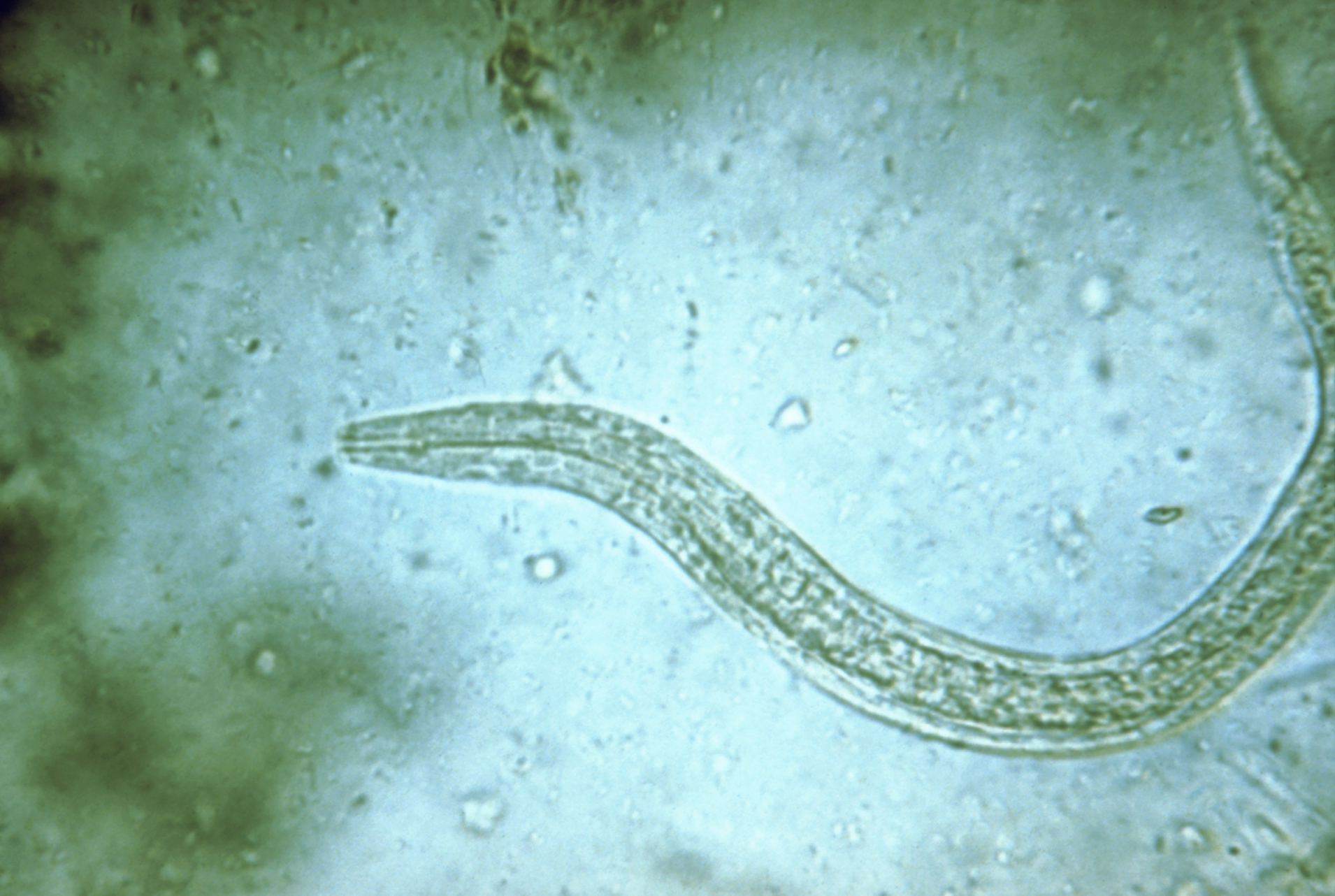
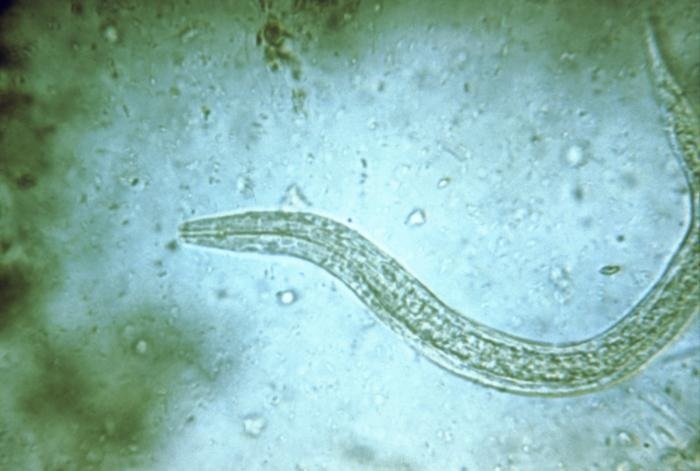
The parasitic hookworm Necator americanus—the most common type that causes infections in humans—is typically passed from the feces of an infected person to another, usually by walking barefoot in contaminated soil. Hookworms can live in the small intestine for years and survive by consuming their host’s blood.
Hookworm is currently treated with annual, widespread administration of anti-worm drugs, which does little to prevent re-infection and may create drug-resistance in the future. A vaccine would allow the body to block hookworms before they attack and improve quality of life for people living in endemic areas like South America, sub-Saharan Africa and Southeast Asia, Dr. Diemert said.
“Current treatment provides temporary relief in terms of curing some infections and reducing the burden of others, but since the people are living in a contaminated environment, they quickly get reinfected,” Dr. Diemert said. “That’s why we believe that developing a preventative vaccine would be a much better means of controlling and potentially eliminating this infection.”
The vaccine will be primarily aimed toward children, who are most susceptible to reinfection and adverse health effects.
“So much of the health impact is because of kids being infected while they’re growing up and developing,” Dr. Diemert said. “The plan is for them to be vaccinated when they are young so they don’t get infected at all during childhood, or they don’t get significant infections.”
In 2015 the researchers began asking volunteers to be infected with hookworm larvae to establish a controlled human infection model. After a few months, researchers monitored the volunteers’ symptoms and hookworm egg counts from fecal samples. Then they were given current approved medication for hookworm and their infections disappeared in just a few days.
This data from Phase one trial volunteers helped the team determine that the infection model could be used to test a vaccine candidate’s efficacy. Dr. Diemert said these volunteers played an integral role in the path toward a viable vaccine.
“We wouldn’t be where we are at all if they hadn’t volunteered. Although they’re less glamorous, phase one trials are probably the most integral part of clinical development,” he said. “If you don’t get past phase one, you aren’t getting anywhere. It was really important to us to establish that the model was safe and figure out a dose of hookworm that results in consistent infection.”
Hookworm is virtually nonexistent in the United States. By testing the vaccine in Washington, D.C., where people are not regularly infected with hookworm, the researchers will accelerate efforts to get the vaccine to people who need it most.
After proving the efficacy of the vaccine in D.C., researchers will seek volunteers in Brazil and Africa to test its ability in a less controlled environment. This will require much larger sample sizes, increased expense and more time. During the field-based trial, the patient volunteers are infected naturally in their environments.
Dr. Bethony said to do an efficacy study in a hookworm endemic area the researchers would have to recruit close to 1,200 volunteers. Each volunteer in the field study would need to be participate for about 24 months compared to just a few months for the planned controlled infection trial in the United States. However, this method also poses challenges, he said.
“Since we are testing the vaccine and challenging hookworm naive participants from the United States, we may get an slightly different response when the vaccine is administered in the resource-poor hookworm endemic areas, as many of the people would have already been infected with hookworm, which could affect their response to the vaccine,” he said.
During this phase two, one of the lead vaccine candidates will be tested in combination with different adjuvants. One of the main objectives of the trial is to down-select the best vaccine formulation to take into the field. In addition to the results of the controlled infection part of the study, collaborators in the Department of Microbiology, Immunology, and Tropical Medicine including Imtiaz Khan and Rebecca Lynch, will look into the immune responses that may help to predict who is protected from hookworm after being vaccinated.
If a vaccine candidate proves to prevent infection, Dr. Diemert said, it likely wouldn’t be available on the market until the mid-2020s. Because it is a pediatric vaccine, the researchers will have additional phases of testing to ensure it is safe and effective for children.
“It’s still a ways off,” Dr. Diemert said. “There are some big trials that would have to be done.”